Lantern Pharma (NASDAQ: LTRN), a clinical-stage biopharmaceutical company leveraging artificial intelligence (AI) and machine learning to transform the cost, pace, and timeline of oncology drug discovery and development, today announced a significant advancement towards the development of a diagnostic for its drug candidate LP-184. The diagnostic is currently based on qRT-PCR (quantitative real-time polymerase chain reaction) technology and is focused on quantifying the amount of PTGR1 RNA in patient tumor samples to assess the potential for sensitivity to Lantern’s drug candidate LP-184. The company plans to further develop and validate the assay for its use as a potential tool for patient selection in later stage clinical trials across a broad range of solid tumors that have shown sensitivity to LP-184.
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20240710249401/en/
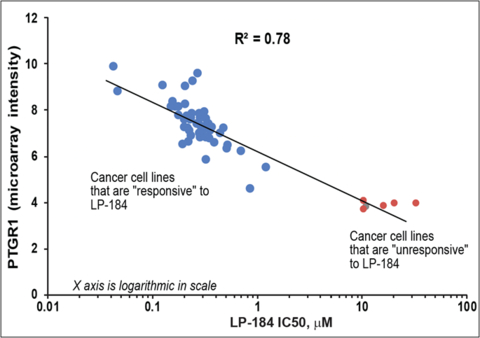
Figure 1: Correlation of PTGR1 expression and cytotoxicity/potency (IC50, µM) of LP-184 (Graphic: Business Wire)
Lantern has successfully confirmed PTGR1 as a key biomarker that it intends to use to optimize patient selection based on potential tumor sensitivity to the drug candidate LP-184. LP-184 is a precision oncology drug candidate with the potential to address multiple solid tumors. This confirmation marks a crucial step towards the development of a companion diagnostic and potential stratification tool to assist with targeted patient selection. Lantern plans on further validations and development of the assay using qRT-PCR and partnering with central labs and cancer centers for eventual use in patient selection and stratification. In a key publication on the utility and value of biomarkers in oncology trials among some of the most common cancers, titled Does biomarker use in oncology improve clinical trial failure risk? A large‐scale analysis by Parker, et al., 2021 in Cancer Medicine found success of clinical trials to be significantly correlated to the incorporation of biomarkers. In particular, the Parker, et al. publication stated that:
“…Our overall analysis of these four cancers, independent of indication, revealed a fivefold benefit of hazard ratios from the Markov models, suggesting a substantial benefit from biomarker use. The hazard ratio analysis of the Markov biomarker models examined how likely clinical trial success was associated with biomarker use versus no biomarker use. Hazard ratios indicated that for biomarker‐based drugs clinical trial success was largest for breast cancer (12‐fold) followed by melanoma (eightfold) and lung cancer (sevenfold) …Our data provide the most extensive look at biomarker use to date in oncology, with an advanced statistical method. Our findings indicate that biomarkers provide a statistically significant benefit, despite the fact our study includes biomarkers not yet FDA approved.”
By incorporating the PTGR1 biomarker into LP-184's development strategy, Lantern Pharma is aligning with best practices in precision medicine and aiming to increase the likelihood of successful clinical outcomes in future clinical trials. PTGR1 levels have been measured to be higher in certain cancer cells than in normal cells, and Lantern is leveraging this biological activity to target the cancer indications believed most likely to respond to drug candidate LP-184. In the October 2023 paper in Molecular Cancer Therapeutics, Lantern along with collaborators from Fox Chase Cancer Center published clear evidence that higher potency of LP-184 (measured in IC50 values) was directly correlated with higher expressions of PTGR1 and that cancer cell lines that did not have PTGR1 expression remained stable in the presence of LP-184 (see figure 1).
"This milestone represents a significant leap forward in our precision oncology approach and in ensuring that we enrich our future LP-184 clinical trials with the patients we believe will be most likely to benefit," said Panna Sharma, CEO of Lantern Pharma. "By working to develop a companion diagnostic for LP-184, we're not just advancing a drug candidate; we're paving the way for more personalized and effective cancer treatments for patients that have the highest likelihood of benefitting from the therapy. The planned use of biomarkers like PTGR1 in our clinical trials exemplifies our commitment to data-driven, patient-centric drug development."
These steps toward development of this companion diagnostic align with Lantern Pharma's commitment to leveraging cutting-edge technology in drug development. By combining AI-driven insights with advanced diagnostic tools, the company aims to accelerate the drug development process and improve patient outcomes.
Lantern Pharma plans to implement this assay in upcoming clinical trials for LP-184, potentially streamlining the development process and increasing the likelihood of successful outcomes. LP-184— a novel therapeutic in clinical development for the potential treatment of malignant gliomas, pancreatic cancer, and atypical teratoid rhabdoid tumors (ATRT)— has also been granted an Orphan Drug Designation by the FDA, along with a Rare Pediatric Disease Designation.
About Lantern Pharma:
Lantern Pharma (NASDAQ: LTRN) is an AI company transforming the cost, pace, and timeline of oncology drug discovery and development. Our proprietary AI and machine learning (ML) platform, RADR®, leverages over 60 billion oncology-focused data points and a library of 200+ advanced ML algorithms to help solve billion-dollar, real-world problems in oncology drug development. By harnessing the power of AI and with input from world-class scientific advisors and collaborators, we have accelerated the development of our growing pipeline of therapies that span multiple cancer indications, including both solid tumors and blood cancers and an antibody-drug conjugate (ADC) program. On average, our newly developed drug programs have been advanced from initial AI insights to first-in-human clinical trials in 2-3 years and at approximately $1.0 - 2.5 million per program.
Our lead development programs include a Phase 2 clinical program and multiple Phase 1 clinical trials. We have also established a wholly-owned subsidiary, Starlight Therapeutics, to focus exclusively on the clinical execution of our promising therapies for CNS and brain cancers, many of which have no effective treatment options. Our AI-driven pipeline of innovative product candidates is estimated to have a combined annual market potential of over $15 billion USD and have the potential to provide life-changing therapies to hundreds of thousands of cancer patients across the world.
Please find more information at:
- Website: www.lanternpharma.com
- LinkedIn: https://www.linkedin.com/company/lanternpharma/
- X: @lanternpharma
Forward-looking Statements:
This press release contains forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as amended. These forward-looking statements include, among other things, statements relating to: future events or our future financial performance; the potential advantages of our RADR® platform in identifying drug candidates and patient populations that are likely to respond to a drug candidate; our strategic plans to advance the development of our drug candidates and antibody drug conjugate (ADC) development program; estimates regarding the development timing for our drug candidates and ADC development program; expectations and estimates regarding clinical trial timing and patient enrollment; our research and development efforts of our internal drug discovery programs and the utilization of our RADR® platform to streamline the drug development process; our intention to leverage artificial intelligence, machine learning and genomic data to streamline and transform the pace, risk and cost of oncology drug discovery and development and to identify patient populations that would likely respond to a drug candidate; estimates regarding patient populations, potential markets and potential market sizes; sales estimates for our drug candidates and our plans to discover and develop drug candidates and to maximize their commercial potential by advancing such drug candidates ourselves or in collaboration with others. Any statements that are not statements of historical fact (including, without limitation, statements that use words such as "anticipate," "believe," "contemplate," "could," "estimate," "expect," "intend," "seek," "may," "might," "plan," "potential," "predict," "project," "target," “model,” "objective," "aim," "upcoming," "should," "will," "would," or the negative of these words or other similar expressions) should be considered forward-looking statements. There are a number of important factors that could cause our actual results to differ materially from those indicated by the forward-looking statements, such as (i) the risk that our research and the research of our collaborators may not be successful, (ii) the risk that promising observations in preclinical studies do not ensure that later studies and development will be successful, (iii) the risk that we may not be successful in licensing potential candidates or in completing potential partnerships and collaborations, (iv) the risk that none of our product candidates has received FDA marketing approval, and we may not be able to successfully initiate, conduct, or conclude clinical testing for or obtain marketing approval for our product candidates, (v) the risk that no drug product based on our proprietary RADR® AI platform has received FDA marketing approval or otherwise been incorporated into a commercial product, and (vi) those other factors set forth in the Risk Factors section in our Annual Report on Form 10-K for the year ended December 31, 2023, filed with the Securities and Exchange Commission on March 18, 2024. You may access our Annual Report on Form 10-K for the year ended December 31, 2023 under the investor SEC filings tab of our website at www.lanternpharma.com or on the SEC's website at www.sec.gov. Given these risks and uncertainties, we can give no assurances that our forward-looking statements will prove to be accurate, or that any other results or events projected or contemplated by our forward-looking statements will in fact occur, and we caution investors not to place undue reliance on these statements. All forward-looking statements in this press release represent our judgment as of the date hereof, and, except as otherwise required by law, we disclaim any obligation to update any forward-looking statements to conform the statement to actual results or changes in our expectations.
View source version on businesswire.com: https://www.businesswire.com/news/home/20240710249401/en/
Contacts
Investor Relations
mailto: ir@lanternpharma.com
ph: (972) 277-1136





